In nutraceutical markets, packaging is not the final step – it is the point where compliance, traceability, and brand credibility are tested. For spirulina producers supplying global buyers, packaging units must be designed to withstand audits as rigorously as cultivation systems withstand biological stress.
The Sea Pearl Nutraceuticals project illustrates how an audit-oriented packaging unit can support export-ready spirulina products, enabling value addition, compliant labelling, and seamless acceptance across international markets.
Client Background: Nutraceutical-Grade Spirulina Supplier
Sea Pearl Nutraceuticals Ltd, also known as Sea Pearl Biotech, is an India-based producer and supplier of spirulina and algae-derived nutraceutical ingredients. Serving health supplement, functional nutrition, and wellness markets, the company operates with a strong emphasis on purity, nutritional integrity, and consistency.
Its operations in the Gandhinagar–Ahmedabad region of Gujarat support large-scale cultivation and processing aligned with nutraceutical industry expectations.
Why Packaging Determines Audit Readiness
For nutraceutical brands, audits extend beyond farming practices into how products are blended, packed, labelled, and documented. Even high-quality spirulina can fail market entry if packaging workflows lack traceability or compliance clarity.
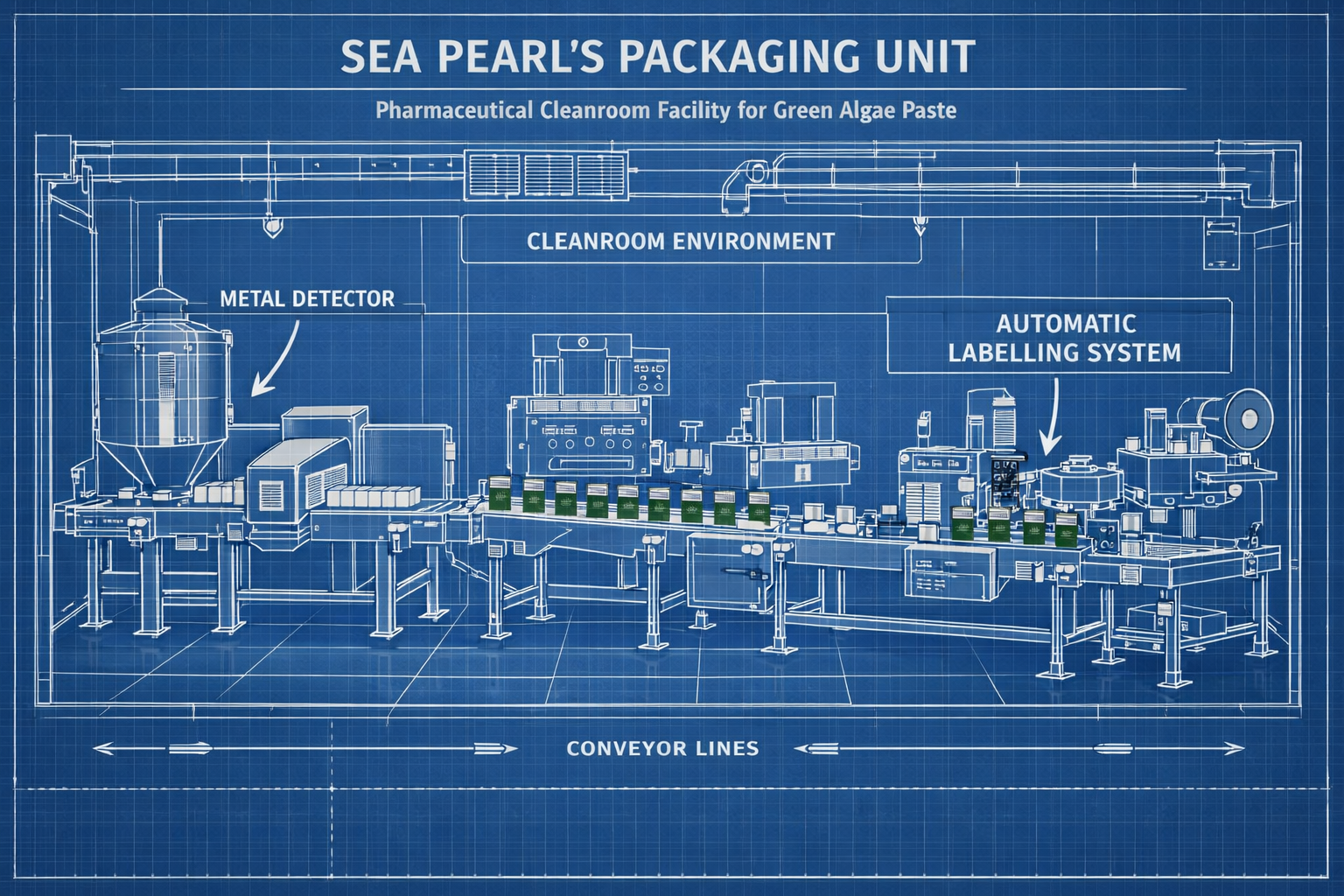
Sea Pearl’s packaging unit was therefore conceptualised not merely as a packing facility, but as an audit-facing system designed to satisfy regulatory scrutiny from day one.
Export-Grade Packaging Infrastructure
The packaging unit operates with clearly defined hygienic zones, controlled material movement, and documentation-ready workflows. This structure supports both retail and bulk export formats while maintaining food-grade handling standards.
Advanced packing systems were aligned with nutraceutical requirements, enabling scalable formats without compromising batch integrity or hygiene.
Value Addition for Global Brands
Beyond basic packing, the Sea Pearl facility supports value-added operations essential for modern nutraceutical products. Controlled blending, precise portioning, and format flexibility allow spirulina to be delivered in consumer-ready forms such as sachets, capsules, and instant mixes.
These workflows preserve spirulina’s nutritional profile while improving shelf stability and usability – key factors for international retail acceptance.
Batch Traceability Built Into the Flow
Audit readiness depends on the ability to trace every finished pack back to its raw material source. Sea Pearl’s packaging unit embeds traceability into daily operations through structured batch handling, record-keeping, and process transparency.
Each stage – from receipt of spirulina to final labelling – is designed to minimise ambiguity and support rapid audit verification.
USDA & Global Labelling Compliance
Labelling systems at Sea Pearl are structured to meet USDA Organic and international export norms. Labels include clear ingredient declarations, nutritional panels, batch identification, manufacturing and expiry details, and origin information.
This audit-designed labelling framework reduces friction during third-party inspections and import clearances, supporting smoother global distribution.
Strategic Advantage for Partner Brands
By leveraging Sea Pearl’s packaging unit, nutraceutical brands such as Gluck gain access to a turnkey pathway for value addition, compliant packing, and export-ready labelling.
This integration allows brand owners to focus on product development and market expansion while relying on an infrastructure designed specifically for global audits.
Greenbubble’s Role in Audit-Oriented Design
Greenbubble Algalworks supported Sea Pearl Nutraceuticals through solution-driven spirulina farming and processing expertise. By aligning cultivation systems with downstream packaging and audit requirements, Greenbubble helped ensure that production quality translated seamlessly into compliant finished goods.
This project highlights Greenbubble’s ability to support nutraceutical-focused producers where audit readiness is a core operational requirement.
What Nutraceutical Producers Can Learn
Key insights from the Sea Pearl project include:
- Packaging units must be designed for audits, not retrofitted later
- Value addition increases compliance complexity and must be controlled
- Traceability and labelling are as critical as cultivation quality
- Export readiness depends on system transparency
Audit success is engineered – not improvised.
FAQs
Q1. Why is packaging critical for global nutraceutical audits?
Packaging is where traceability, hygiene, and labelling are verified by regulators and buyers.
Q2. Can one packaging unit support multiple brands?
Yes. With structured workflows and documentation, a single unit can serve multiple compliant brands.
Q3. Does value addition increase audit complexity?
Yes. Blending and portioning require stricter control but can be managed through disciplined design.
Q4. How does labelling affect export acceptance?
Correct labelling ensures regulatory compliance and smooth import clearance.
Q5. Can existing facilities be upgraded for audit readiness?
Yes. With proper flow audits and system redesign, audit readiness can be retrofitted.
Conclusion: Packaging as an Audit Interface
The Sea Pearl Nutraceuticals packaging unit demonstrates that global audit readiness is achieved through deliberate design. By integrating hygienic infrastructure, traceability, compliant labelling, and value-added workflows, the facility functions as a reliable interface between spirulina production and international markets. With Greenbubble’s support, packaging becomes not a vulnerability – but a strategic advantage.